Batteries & Energy Storage
COMMON CHALLENGES
Modern energy storage technologies, such as lithium-ion batteries, are used in more and more applications every day. The proliferation of energy storage technologies also drives the demand for materials with improved performance in key metrics such as power and energy density, Coulombic efficiency, cycle lifetime, safety, and stability.
Researchers can develop new materials or chemistries to improve on key performance metrics by understanding how they are affected by fundamental aspects like crystal structure, diffusion rates, electron transport, and other electrochemical dynamics. Characterizing materials properties, such as atomic structure and elemental distribution, is the first step towards elucidating these structure-property relationships.
INNOVATIVE TECHNIQUES
Several techniques are available to better characterize and understand crucial structure-property relationships in energy storage materials.
|
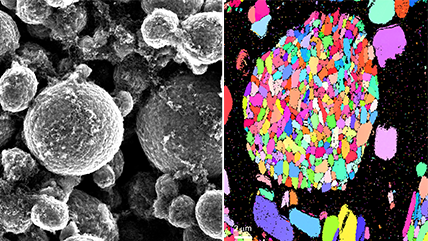
Electron backscatter diffraction (EBSD) Allows detailed microstructural analysis by mapping crystal orientations, phases, and grain boundaries, helping researchers understand material properties, deformation, and failure mechanisms at the microscale. |
Electron energy loss spectroscopy (EELS) Delivers atomic-scale insights into elemental composition and chemical bonding—enabling a deeper understanding of material properties to advance fundamental research and discovery. Includes energy-filtered TEM (EFTEM). Elevate your EELS at EELS.info. |
|
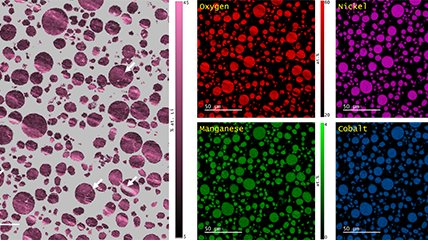
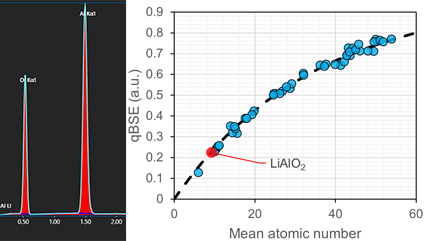
Energy dispersive x-ray spectroscopy (EDS/EDX) Facilitates rapid, reliable elemental analysis by detecting characteristic x-rays emitted from a sample, allowing researchers to identify and quantify major, minor, and trace elements across a wide range of materials. |
Integrates EELS, EDS, 4D STEM, and more to deliver rich, correlated insights—advancing the understanding of complex, dynamic nanoscale phenomena. |
|
Delivers high-resolution visualization of biological and inorganic specimens, enabling researchers to study ultrastructure, material growth, and failure mechanisms with exceptional clarity. |
Captures real-time nanoscale dynamics under controlled stimuli to uncover fundamental mechanisms and accelerate scientific discovery. |
|
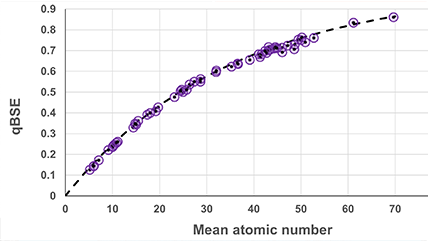
Integrated SEM Combines EDS, EBSD, CL, BSE, and WDS into unified platforms—delivering comprehensive, high-resolution insights that streamline workflows and deepen understanding of complex materials. |
Micro x-ray fluorescence (micro-XRF) Delivers high-resolution, non-destructive elemental analysis—ideal for layered, sensitive, or irregular samples. |
|
Wavelength dispersive x-ray spectroscopy (WDS) Offers high spectral resolution and sensitivity, enabling precise identification and quantification of trace and light elements in complex samples—especially where overlapping x-ray lines challenge other techniques. |
Visit solar, utilities, and environment and semiconductor materials and devices to learn about related applications.
ENABLING RESULTS
Many initial studies on new energy storage materials focus on characterizing material properties in the pristine state, followed by determining property changes after cycling to different states of charge, e.g., ex-situ characterization. Electron microscopy provides various imaging and elemental analysis techniques with micro-scale to atomic-scale spatial resolution.
The choice of scanning electron microscopy (SEM) versus transmission electron microscopy (TEM) techniques depends on the nature of the specimen (e.g., bulk, powder, nanomaterial) and the desired analysis.
Common SEM characterization
- Surface imaging of an electrode over a large region of interest
- Imaging cross-sections of an electrode or battery cell
- Elemental analysis over a large region of interest with EDS
- Grain structure and orientation analysis of cross-sections with EBSD
Applications
Common TEM characterization
- Nanometer and atomic-scale imaging of materials
- Crystal structure determination with electron diffraction techniques (e.g., selected area electron diffraction, diffraction tomography, 4D STEM)
- Elemental analysis with EDS and EELS
- Chemical state analysis of individual elements with EELS
- Imaging of elemental or chemical state distribution with EFTEM


Energy storage materials exist in constantly evolving (electro)chemical states as they repeatedly convert chemical energy into electrical energy and vice-versa. Understanding this dynamic behavior is key to developing high-performance materials. Studies of this type are accomplished in an electron microscope by imaging a specimen or conducting chemical analysis while also providing an electrochemical stimulus to the specimen, e.g., in-situ microscopy.
There are various setups for in-situ TEM experiments on energy storage materials. Most experiments choose to study an individual element of an entire energy storage device, e.g., the cathode of a battery or a particular half-cell reaction. Simpler experiments forgo using a liquid electrolyte in the in-situ experiment to avoid the complication and still observe dynamic phenomena, at the cost of being a degree removed from actual battery operation conditions. More complex experiments use a specialized TEM specimen holder to bring a full liquid electrolyte cell into the TEM.
In either case, in-situ experiments present several challenges to the microscopist in addition to the basic experimental design and setup:
- Working under low-dose conditions to prevent specimen damage and other beam-induced artifacts
- Acquiring data with the required spatial and temporal resolution to study the reactions of interest properly
- Managing the large amount of data generated (in many cases, several TBs of video data) during a single in-situ experiment
Gatan’s portfolio of in-situ products greatly aids microscopists in addressing and managing all the challenges that come with in-situ experiments on energy storage materials.
Applications
|
Lithium quantification using the composition-by-difference method |
Imaging a lithium metal battery solid electrolyte interphase |
|
|
Observing beam-induced dendritic growth over two different timescales |
|
|
The success of any microscopy experiment begins with proper specimen preparation. The best technique for the material depends on the intended analysis (e.g., TEM vs. SEM analysis), specimen morphology (e.g., powder material vs. assembled coin cell), material sensitivity, among other factors. Critical to the specimen preparation process is the ability to cut, polish, etch the specimen without introducing artifacts that obscure the actual state and morphology of the specimen of interest. Gatan specializes in broad ion beam tools for preparing SEM and TEM specimens rapidly and with minimal artifacts from the preparation process.
SEM preparation
Surface analysis of an electrode or similar material can be straightforward and may only require deposition of a thin conductive layer (such as amorphous carbon or a thin metal layer) to avoid charging during observation in the SEM.
Researchers may be interested in viewing the interior structure of their electrode or cell assembly, and in those cases, cross-section preparation is necessary. Gatan’s broad argon ion beam tools can prepare cross-section specimens of larger area than possible with a focused ion beam (FIB) approach and do so through a simpler preparation process with lower cost and faster preparation time.

TEM preparation
If the material of interest is a powder, it is often possible to simply disperse the powder onto a TEM grid with some amorphous support (e.g., C, SiO2) and do imaging on thin areas without any extra preparation. In other cases, it may be necessary to prepare site-specific specimens or cross-sections using a FIB. Utilizing the PIPS™ tool as a final step after FIB can remove surface contamination (such as redeposition of milled material from the FIB) or surface oxidation and leave a clean surface for TEM analysis.
Applications
|
Argon ion polishing of focused ion beam specimens in PIPS II system |
Most battery materials and energy storage materials, in general, are relatively air-sensitive. Certain materials can show extreme air-sensitivity in their pristine state (such as Li metal anodes). Incorporating air-free specimen transfer techniques into the entire experiment workflow is a very important step that is easily overlooked. Furthermore, the stability of the material in the microscope itself can be a challenge, as the electron beam can cause unwanted changes or completely destroy the specimen during analysis. There are several available tools from Gatan to assist with appropriately handling air-sensitive and beam-sensitive specimens.
Air-free transfer of SEM specimens
The ILoad™ system provides a specialized specimen transfer pod and airlock system for Gatan specimen preparation tools and an SEM. A specimen can be loaded inside a glovebox and into the transfer pod, allowing the specimen to be safely transported out of the glovebox while remaining in an inert gas atmosphere with positive pressure to prevent air from entering the chamber. The transfer pod interfaces with airlocks on the PECS™ tool in addition to a customized airlock for an SEM.

Air-free transfer of TEM specimens
The ILoad system additionally interfaces with the PIPS tool, allowing you to bring a specimen out of the glovebox and into the PIPS with the same inert gas atmosphere. After specimen preparation, the specimen can be brought back to the glovebox and easily loaded into the model 648 holder for transfer under vacuum or inert gas atmosphere into the TEM.
Cryo-transfer of TEM specimens
An increasingly popular technique for studying beam-sensitive materials is the use of cryo-transfer holders to freeze the specimen at cryogenic temperatures for observation. Using a cryo-transfer holder minimizes unwanted surface reactions that may happen at room temperature, and also improves specimen stability under the electron beam.
Applications
|
Imaging discrete ions at a liquid-solid interface using low-dose cryo-EM and electron counting |