The importance of the specimen support film for cryo-TEM
Holey carbon films, used with or without an additional overlay of thin (~ 5 nm) carbon, are a commonly used specimen support for cryo-TEM studies of biological macromolecules. These holey support films can be made in the laboratory, or they can be purchased commercially. The preparation methods for most homemade holey supports involve casting a thin layer of resin such as polyvinyl formal (commercially known as Formvar® or Vinylec®), which is then further treated to produce the perforations. The perforated film is then applied to the surface of an electron microscope grid and a layer of carbon is evaporated onto the surface to enhance its stability and conductivity when exposed to the electron beam of the TEM. The resin layer is often removed by exposing it to solvent with the intent of leaving just the holey carbon layer. Beside the amount of time and the multiple steps required to prepare these support films, it is difficult to get reproducible results with regard to overall film thickness, hole size or hole spacing. Although somewhat expensive, commercially prepared films are an attractive alternative to homemade films because they are fabricated with a specified thickness, planarity, and hole shape, size and spacing all with minimal variation from one batch to the next.

A common aspect of support films, whether homemade or commercially produced, is the variability of the surface charge. Although there are some exceptions such as certain membrane proteins, the majority of biological macromolecules prefer a hydrophilic surface. The visual indication of what occurs as the solution that contains the specimen is applied to the surface of the support film is an important qualitative measure of the surface charge. Ideally one wants this specimen solution, which is usually anywhere from 2 – 5 μL, to spread out evenly across the surface such that the majority of the specimen particles partition within the holes of the support without causing any changes to the macromolecular structure of the specimen. If the specimen solution ‘beads up’ when applied to the support film, the amount of specimen that actually partitions to the holes may be very small, thus limiting the amount of data that can be gathered.
Developing a reproducible means for preparing the surface charge of the support film prior to applying the specimen can improve the quality of the cryo-preparation and maximize the amount of data that can be gathered from each specimen grid. To render specimen supports hydrophilic, plasma cleaning is now widely accepted as the method of choice. Commercial instruments designed for this purpose basically consist of a low vacuum chamber to contain the item to be cleaned and a means to introduce specific mixtures of gases that can be ionized by an electrical current to form the plasma. Absorption of electrical energy by the gas increases its temperature causing the ions to increase speed. These energetic ions can then interact with the exposed surfaces of the items being cleaned. Ion bombardment by an inert gas, such as argon, is similar to sandblasting; small amounts of material will be removed from the surface. When a gas mixture containing oxygen is used, bombardment of the surface causes chemical reactions that result in the removal of hydrocarbon contaminants from the surfaces of the items being cleaned. When cleaning hydrocarbon contamination from the surface of fragile holey carbon support films, it is important that the plasma is gentle enough such that it does not break the support during the cleaning process. As a rule of thumb, the specimen supports should be kept in a clean environment and be used within one hour following plasma cleaning.

The Solarus® advanced plasma cleaning system is a state-of-the-art, versatile and easy to use plasma cleaner. It provides optimal plasma power for any cleaning condition by incorporating a real-time radio frequency (RF) auto-matching network and a variable RF power supply operating at 10 to 50 W. Mass flow controllers (MFC) for hydrogen, oxygen and argon gas automatically provide accurate gas flow to the cleaning chamber ensuring stable plasma production during the cleaning process. The unique H2/O2 gas mixture recipe (patent pending) provides superior cleaning with less sputter damage for even the most fragile specimens, and the plasma runs approximately 50% cooler than that from gas mixtures typically used such as Ar or Ar/O2. Using the interactive touch-screen interface, every user can obtain consistent results by selecting one of seven factory programmed recipes for which the processing parameters have been optimized based on the particular type of specimen being cleaned. The spacious top entry specimen chamber can accommodate 40 to 50 specimen support films at one time or two TEM specimen holders, the latter of which can be inserted through ports at the front of the chamber. Process cycle rates of less than two minutes ensure high throughput performance.

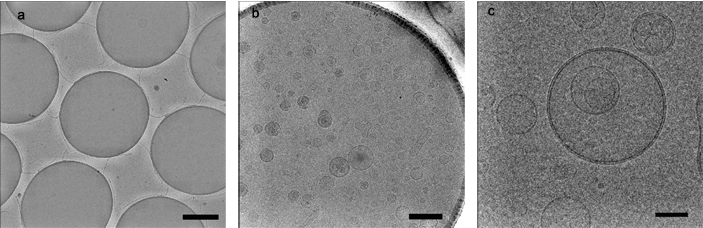
Using a reproducible method for cleaning holey carbon support films is important because it improves the even spreading of the specimen solution over the surface of the support. With its proprietary H2/O2 gas mixture recipe, the Solarus Advanced Plasma Cleaning System can improve the surface characteristics for holey carbon support films by gently and efficiently removing hydrocarbon contamination, resulting in more consistent and more uniform specimen distribution in cryo preparations and therefore more productive data collection sessions at the electron microscope.