Nakamura Group: University of Tokyo
"Scientists should provide solutions to social problems and dreams for people."
The Nakamura Group is tackling this goal by means of physical organic chemistry and synthetic chemistry.
Lab Focus
It has long been the dream of chemists to elucidate the intrinsic nature of atoms, molecules, and their assemblies, together with controlling their functions. We are exploring new concepts of structural chemistry and the creation of new functional materials using original synthetic tools developed by ourselves. We are also promoting research into the creation, property control, and biological function of molecular assemblies at angstrom or nanoscale level, and even above (nano-integration). This research is based on fundamental investigations into the structure and reactivity of organic molecules, using various tools such as high-resolution electron microscopy, scanning probe microscopy, and theoretical calculations, together with the development of powerful synthetic methods. These studies are supported by research grants from the Ministry of Education, Culture, Sports, Science, and Technology, with the ultimate goal to reach for scientific dreams, to sustain a healthy life, and to address environmental issues and resource depletion.
Recent News
Real-time Video Imaging of Mechanical Motions of a Single Molecular Shuttle and Sub-millisecond Sub-angstrom Precision
Chemistry in motion: Researchers produce first-ever videos of chemical synthesis at atomic resolution
Research Projects
A scientists' dream: "Imaging single organic molecules in motion" is becoming a reality

Since the concept of atoms and molecules was established a long time ago, it has been the dream of scientists to observe the motion of single small organic molecules. We showed in a 2007 Science paper1 that this is no longer a dream, by studying using a high-resolution electron microscope to in-situ record movies the motions of small organic molecules trapped in or attached to carbon nanotubes2. Since then, we reported movies of the conformational changes of a number of small molecules3 and even performed a kinetic analysis of chemical reactions by analyzing individual reaction events one by one by recording chemical events continuously by using a CMOS camera (Gatan OneView)4,5.
We can also study for the aggregation of single organic molecules leading to nucleation and crystal growth6. More recently, we have succeeded in isolating minute reaction intermediates at each stage in metal-organic framework synthesis and seeing how these changed with time7.

 Once considered too futuristic, this research is seeing rapid development owing to a state-of-the-art atomic resolution electron microscope in full operation in the basement of the Molecular Life Innovation building where our lab is located.
Once considered too futuristic, this research is seeing rapid development owing to a state-of-the-art atomic resolution electron microscope in full operation in the basement of the Molecular Life Innovation building where our lab is located.
Functional carbon clusters: Synthesis, assembly, function, and applications
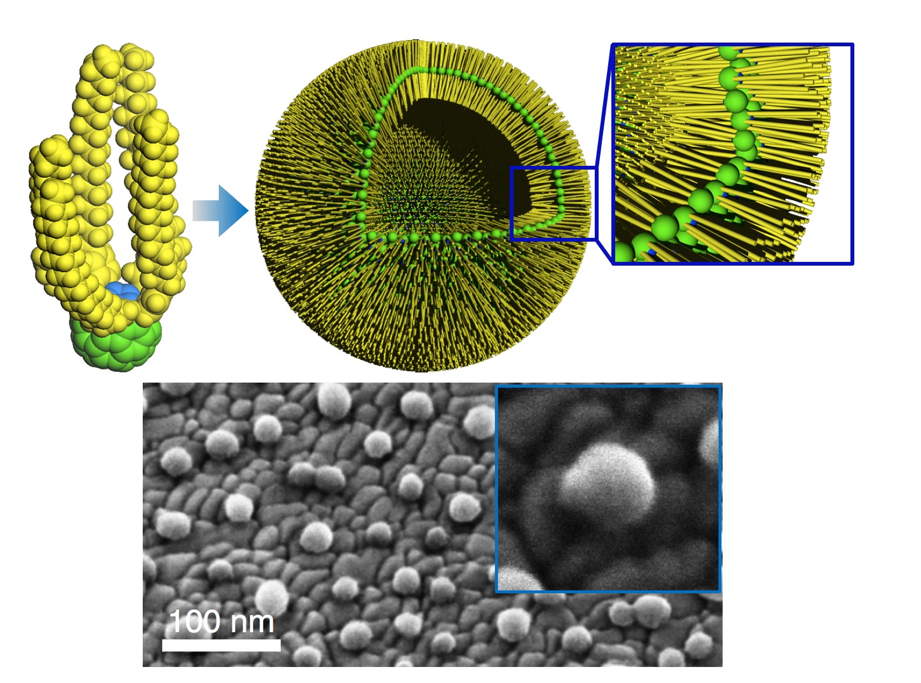
π-Conjugated organic compounds possess unique electronic properties and assemble to form functional materials. We have developed new carbon-rich p-conjugated materials (functional carbon clusters) and investigated their materials and biological applications. For example, when nanometer-sized [60]fullerene molecules are functionalized and aligned horizontally, water-tight fullerene bilayer membranes form and assemble into nanoscale vesicles8. We also achieved DNA and siRNA delivery to animals using an aminofullerene carrier9. In addition to these curved π-electron compounds, we have developed a rigid planar π-electron system, carbon-bridged oligo(p-phenylenevinylene)s (COPVs), which serves as a molecular wire that transfers electrons very fast10. A series of COPV compounds are also applicable to thin-film organic lasers with long operational lifetimes and wavelength tunability11.
Optoelectronic devices for solving energy issues: Contributions of synthetic organic chemists to new materials design
Solar cell manufactured using a low-temperature solution process provides a promising solution to the energy problems in modern society. We have conducted fundamental research and practical applications of "organic thin-film solar cells" and "organic-inorganic hybrid solar cells" in the past 15 years. For example, with the understanding of the formation mechanism of lead perovskite materials12, we developed a polymer stabilization method for perovskite device fabrication13 and tailor-designed new types of a hole transporting materials to obtain perovskite devices with high operational stability and power conversion efficiency14. We also designed and synthesized a new fluorinated cyclic phosphate solvent for use in safe lithium-ion batteries with high energy densities15. We utilize our expertise in microscopy to elucidate the mechanism of materials organization and also explore modern organic synthesis (catalysis) as a tool to create new conjugated molecules for materials science. We maintain close collaboration with companies in this research.

“Element Strategy Initiative”: Organic synthesis using ubiquitous metals
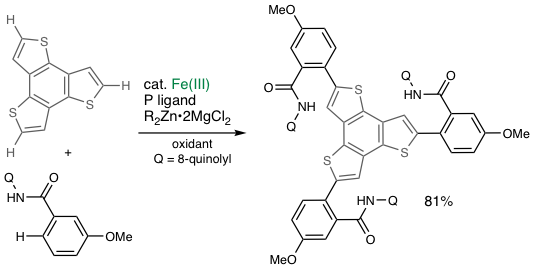
Discoveries made by Noyori, Suzuki, and Negishi et al. in the 1980s exploring precious metal complexes (Ru, Pd) as catalysts for organic synthesis were awarded the Nobel prize. As early as in the late 1990s, we started to focus on "ubiquitous metal catalysis" using iron, cobalt, manganese, and chromium16 and proposed a national policy of “Elemental Strategy Initiative," now broadly recognized as a part of sustainable development goals (SDGs). We recently reached a new stage of our research by achieving iron-catalyzed two-fold C−H activation/C−C coupling with 100% selectivity for cross-coupling17. We recently developed an iron-catalyzed asymmetric synthesis of a proton pump inhibitor drug in collaboration with a company.
Recent Publications
- Imaging Single Molecules in Motion, M. Koshino, T. Tanaka, N. Solin, K. Suenaga, H. Isobe, and E. Nakamura, Science, 316, 853 (2007)
- Atomic-Resolution Transmission Electron Microscopic Movies for Study of Organic Molecules, Assemblies, and Reactions: The First 10 Years of Development, E. Nakamura, Acc. Chem. Res., 50, 1281–1292 (2017).
- Electron Microscopic Observation of Selective Excitation of Conformational Change of a Single Organic Molecule, R. M. Gorgoll, E. Yücelen, A. Kumamoto, N. Shibata, K. Harano, E. Nakamura, J. Am. Chem. Soc., 137, 3474-3477 (2015).
- Direct Microscopic Analysis of Individual C60 Dimerization Events: Kinetics and Mechanisms, S. Okada, S. Kowashi, L. Schweighauser, K. Yamanouchi, K. Harano, E. Nakamura, J. Am. Chem. Soc., 139, 18281-18287 (2017).
- Chemical Kinetics Study through Observation of Individual Reaction Events with Atomic-Resolution Electron Microscopy, E. Nakamura, K. Harano, Proc. Jpn. Acad., Ser. B, 94, 428−440 (2018).
- Heterogeneous Nucleation of Organic Crystals Mediated by Single-Molecule Templates, K. Harano, T. Homma, Y. Niimi, M. Koshino, K. Suenaga, L. Leibler, E. Nakamura, Nat. Mater., 11, 877-881 (2012).
- Atomistic Structures and Dynamics of Prenucleation Clusters in MOF-2 and MOF-5 Syntheses, J. Xing, L. Schweighauser, S. Okada, K. Harano, E. Nakamura, Nat. Commun., 10, 3608 (2019).
- Neutron Scattering Reveals Water Confined in a Watertight Bilayer Vesicle, W. Abuillan, A. S. Becker, B. Demé, T. Homma, H. Isobe, K. Harano, E. Nakamura, M. Tanaka, J. Am. Chem. Soc., 140, 11261–11266 (2018).
- In Vivo Gene Delivery by Cationic Tetraamino Fullerene, R. Maeda-Mamiya, E. Noiri, H. Isobe, W. Nakanishi, K. Okamoto, K. Doi, T. Sugaya, T. Izumi, T. Homma, E. Nakamura, Proc. Natl. Acad. Sci. USA, 107, 5339-5344 (2010).
- Electron transfer through rigid organic molecular wires enhanced by electronic and electron–vibration coupling, J. Sukegawa, C. Schubert, X. Zhu, H. Tsuji, D. M. Guldi, E. Nakamura, Nat. Chem., 6, 899–905 (2014).
- Carbon-bridged oligo(p-phenylenevinylene)s for photostable and broadly tunable, solution-processable thin film organic lasers, M. Morales-Vidal, P. G. Boj, J. M. Villalvilla, J. A. Quintana, Q. Yan, N.-T. Lin, X. Zhu, N. Ruangsupapichat, J. Casado, H. Tsuji, E. Nakamura, M. A. Díaz-García, Nat. Commun., 6, 8458 (2015).
- Chemical Pathways Connecting Lead(II) Iodide and Perovskite via Polymeric Plumbate(II) Fiber, Y. Guo, K. Shoyama, W. Sato, Y. Matsuo, K. Inoue, K. Harano, C. Liu, H. Tanaka, E. Nakamura, J. Am. Chem. Soc., 137, 15907-15914 (2015).
- Polymer Stabilization of Lead(II) Perovskite Cubic Nanocrystals for Semitransparent Solar Cells, Y. Guo, K. Shoyama, W. Sato, E. Nakamura, Adv. Energy Mater., 6, 1502317 (2016).
- Disodium Benzodipyrrole Sulfonate as Neutral Hole-Transporting Materials for Perovskite Solar Cells, R. Shang, Z. Zhou, H. Nishioka, H. Halim, S. Furukawa, I. Takei, N. Ninomiya, E. Nakamura, J. Am. Chem. Soc., 140, 5018-5022 (2018).
- A Cyclic Phosphate-based Battery Electrolyte for High-voltage and Safe Operation, Q. Zheng, Y. Yamada, R. Shang, S. Ko, Y. -Y. Lee, K. Kim, E. Nakamura, A. Yamada, Nat. Energy, DOI: 10.1038/s41560-020-0567-z.
- Iron-Catalyzed C-H Bond Activation, R. Shang, L. Ilies, E. Nakamura, Chem. Rev., 117, 9086–9139 (2017).
- Homocoupling-free Iron-catalysed Twofold C–H Activation/Cross-couplings of Aromatics via Transient Connection of Reactants, T. Doba, T. Matsubara, L. Ilies, R. Shang, E. Nakamura, Nat. Catal. 2, 400-406 (2019).

